īoron also has a characteristic green color when ignited, so it is commonly used in pyrotechnics and flares. Boric acid (H 3BO 3) is used in insulation as a flame retardant. The most important compound of boron is known as sodium borate pentahydrate, used to manufacture fibreglass insulation and bleach. Boric oxide is used to manufacture tough, heat resistant glass known as Pyrex. Several compounds of boron are in common use commercially. This also makes it desirable as a radiation shield, as well as a neutron detector. This makes it useful in the control rods of nuclear reactors to maintain a safe nuclear chain reaction. Communications Chemistry 5, Article number: 149 ( 2022 ) Cite this article 5292 Accesses 17 Citations 4 Altmetric Metrics Abstract The subclass of multi resonant thermally activated delayed. Some useful atomic properties of boron include: Atomic weightĭue to boron's high neutron cross section it is an effective neutron absorber. It is not found in high abundance in the universe or on Earth, however there is enough of it on Earth to satisfy its certain applications in human society. 
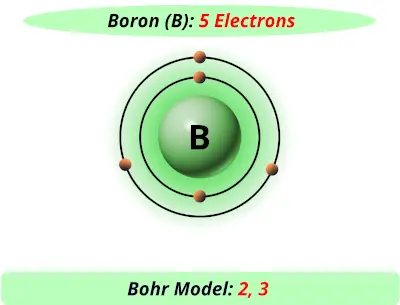
īoron ( B) is the 5 th element on the periodic table. Boron, atomic weight of 10.811 and atomic number of 5. Therefore, there are various non-equivalent definitions of atomic radius.Figure 1. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. Gun Complete W/5 Boron Nozzle 5/16 Diameter SKU: 20-043-5 Description Your browser cant play this video. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Boron atom is 84pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. Benefits Dosage and how to take Side effects Other supplements for arthritis Summary Research suggests that boron may be an effective way to treat the symptoms of rheumatoid arthritis and. 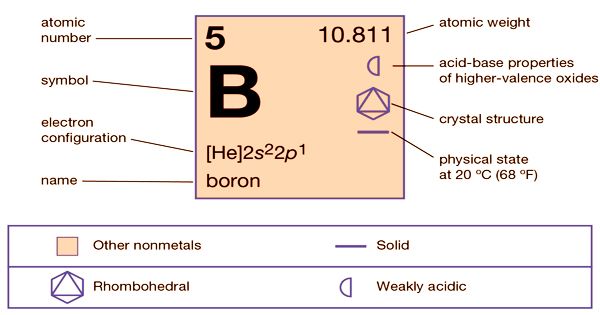
The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Boron are 10 11. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Atomic Number – Protons, Electrons and Neutrons in Boronīoron is a chemical element with atomic number 5 which means there are 5 protons in its nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
